Required Background Knowledge:
|
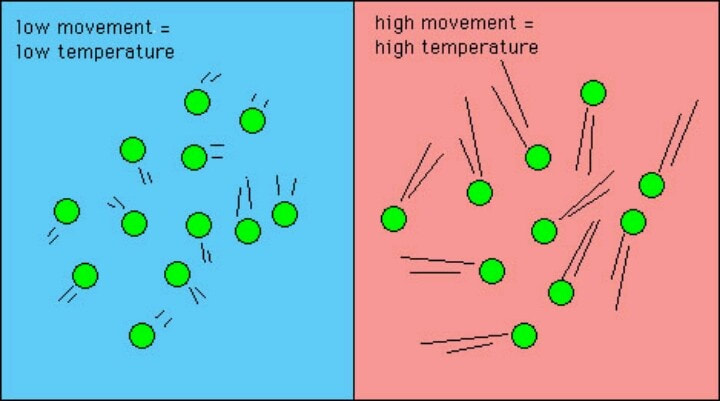
Heat is a form of energy. As such, heat is not the same thing as temperature. In order to understand how heat impacts temperature, it is first important to understand how heat moves from one place to another. When heat is added, particles in a substance begin to move faster and move closer together, ultimately resulting in a higher temperature. Contrarily, when heat escapes, particles slow down and spread further apart, decreasing the temperature.
An effective seal is paramount to maintaining the heat of a liquid. When a solid seal is in place, heat has little room to escape which allows the temperature of a liquid inside a container to stay constant. This is the case for both hot and cold liquids. |
Beyond the seal, good bottle design comes down to the concept of insulation. Certain materials do a better job of ensuring that heat cannot pass through it. Plastic is a great insulator, but as students will find in this design challenge, it poses a unique set of problems, mainly with regards to ensuring that it can withstand increased heat and temperature without structural collapse. Stainless steel, which has become the industry standard for a wide variety of thermal mugs can keep liquids hot or cold for long periods of time, but run the risk of being too hot to touch. Innovative solutions like a double walled designs can circumvent this issue and are a beacon for the kinds of ideas that can come out of this project.
|